GLP-2 T 30MG
$130.00MG
Overview:
Tirzepatide is a dual agonist of the GLP-1 and GIP receptors for metabolic modulation.
Details:
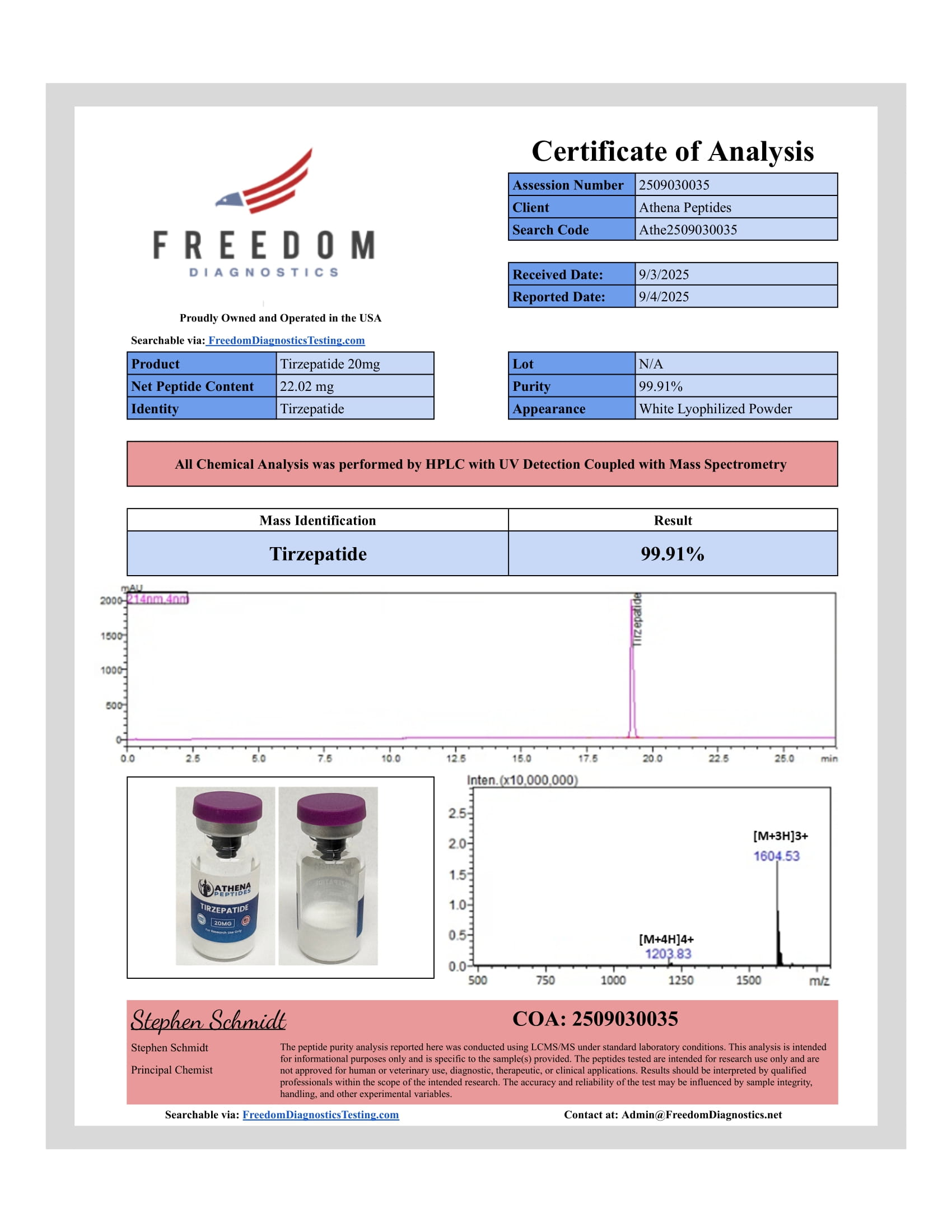
- Purity: ≥ 98% (HPLC)
- Molecular Formula: C225H348N48O68
- Molecular Weight: 4817.5 g/mol
- Form: Lyophilized powder
- Storage: –20°C, protected from light and moisture
Intended Use:
For research purposes only. Not approved for human or veterinary use.
In stock
Overview
GLP-2(T) represents a paradigm shift in dual incretin receptor pharmacology, functioning as an engineered 39-amino acid peptide with balanced GIP receptor agonism coupled to biased GLP-1 receptor activation. Unlike single-target incretin mimetics, this chimeric construct exploits synergistic GIP/GLP-1R co-activation to drive superior glucose-dependent insulin secretion while simultaneously modulating central appetite circuits and peripheral energy expenditure pathways.
The molecule’s structural architecture—a GIP backbone with C20 fatty diacid lipidation—confers albumin binding properties that extend plasma half-life to approximately 5 days, enabling once-weekly subcutaneous dosing kinetics. This pharmacokinetic profile, combined with dual-receptor signaling bias toward cAMP generation over β-arrestin recruitment at the GLP-1R, produces metabolic efficacy surpassing selective GLP-1 agonists in head-to-head clinical comparisons.
Key Characteristics
MOLECULAR PROFILE
- Formula: C225H348N60O68
- Weight: 5047.61 g/mol
- CAS: 2023788-19-2
- Structure: 39 amino acid GIP-based peptide with C20 lipid modification
- Half-life: ~5 days (albumin-bound)
PHYSICAL PROPERTIES
- Form: White to off-white lyophilized powder
- Solubility: Readily dissolves in water
- Storage: Keep refrigerated 36‑46 °F (2‑8 °C)
- Reconstitution: Mix with bacteriostatic water
Dual Incretin Receptor Mechanisms
GLP-2(T) exploits coordinated GIP and GLP-1 receptor activation to generate supraphysiological incretin responses. The molecule exhibits native GIP pharmacology at GIPR while demonstrating biased agonism at GLP-1R—preferentially activating adenylyl cyclase/cAMP/PKA signaling cascades while minimizing β-arrestin recruitment and receptor internalization.
GIP Receptor Signaling
Pancreatic β-Cell Actions
GIPR activation couples to Gs proteins, triggering adenylyl cyclase to generate cAMP. Elevated cAMP activates PKA and EPAC2, potentiating glucose-dependent insulin granule exocytosis. GIPR signaling is obligatory for GLP-2(T)’s insulinotropic effects in human islets.
Adipocyte Metabolism
GIP receptor engagement in adipose tissue modulates lipid partitioning and energy storage. While traditional models emphasized lipogenic effects, emerging data suggest GIPR activation enhances adipocyte insulin sensitivity and thermogenic capacity under specific metabolic contexts.
Central Nervous System
GIPR expression in hypothalamic appetite-regulating nuclei contributes to GLP-2(T)’s anorectic properties. Dual GIP/GLP-1 signaling in the brain produces synergistic effects on food intake suppression exceeding either pathway alone.
GLP-1 Receptor Signaling
Biased Agonism Profile
GLP-2(T) exhibits signaling bias at GLP-1R, favoring cAMP generation over β-arrestin-1/2 recruitment. This pharmacological profile prevents rapid receptor desensitization and trafficking, sustaining insulin secretory responses. β-arrestin1 normally limits GLP-1-stimulated insulin release; GLP-2(T)’s bias circumvents this negative regulatory mechanism.
Glucagon Suppression
GLP-1R activation in pancreatic α-cells inhibits glucagon secretion via cAMP-mediated pathways distinct from β-cell stimulation. Reduced glucagon output diminishes hepatic glucose production, contributing ~40% of glycemic improvement in type 2 diabetes models.
Gastric Motility Control
GLP-1R engagement in brainstem neurons delays gastric emptying through vagal efferent pathways. Slower nutrient delivery to the small intestine attenuates postprandial glucose excursions and prolongs satiety signaling. This effect demonstrates tachyphylaxis over 20-30 weeks but contributes meaningfully to early metabolic improvements.
SURPASS & SURMOUNT Clinical Programs
The SURPASS (type 2 diabetes) and SURMOUNT (obesity) phase 3 programs enrolled >15,000 participants across 10 randomized controlled trials, establishing GLP-2 (T)’s superiority over selective GLP-1 receptor agonists and basal insulin in head-to-head comparisons.
SURPASS-1: Monotherapy Efficacy
478 treatment-naïve T2D patients randomized to GLP2-T (5/10/15 mg) vs placebo for 40 weeks:
- HbA1c reductions: -1.87% (5 mg), -1.89% (10 mg), -2.07% (15 mg) vs +0.04% placebo
- 87-97% of participants achieved HbA1c <7% (vs 20% placebo)
- Weight loss: -7.0 to -9.5 kg dose-dependent reduction
- Superior glycemic control without background medications
SURPASS-2: Head-to-Head vs Semaglutide
1,879 T2D patients on metformin randomized to GLP-2(T) vs GLP-1 (S) 1 mg for 40 weeks:
- GLP2-T 15 mg reduced HbA1c -2.46% vs -1.86% with GLP1-S (p<0.001)
- Weight loss: -11.2 kg (15 mg) vs -5.7 kg GLP1-S
- 62% achieved dual endpoint (HbA1c <5.7% + ≥5% weight loss) vs 18% GLP1-S
- Established dual-agonist superiority over selective GLP-1RAs
SURMOUNT-1: Obesity Pharmacotherapy
2,539 adults with obesity (BMI ≥30) or overweight (BMI ≥27) with comorbidities, 72 weeks:
- Mean weight reduction: -15.0% (5 mg), -19.5% (10 mg), -20.9% (15 mg) vs -3.1% placebo
- 89% of participants on 10 mg achieved ≥5% weight loss (vs 28% placebo)
- 50% achieved ≥20% weight loss on 10/15 mg doses—unprecedented in pharmacotherapy
- Significant improvements in cardiometabolic markers (BP, lipids, inflammatory markers)
SURMOUNT-2: Obesity + Diabetes
938 adults with obesity/overweight and type 2 diabetes on metformin ± SGLT2i/sulfonylurea:
- Weight loss: -12.8% (10 mg), -14.7% (15 mg) vs -3.2% placebo at 72 weeks
- HbA1c reduction: -2.07% (10 mg), -2.11% (15 mg) vs -0.51% placebo
- Dual benefit in challenging population with metabolic comorbidity
- Sustained effects maintained through study completion
Cardiovascular Risk Reduction
Post-hoc analysis and meta-analysis of cardiovascular outcomes across trials:
- Systolic BP reduction: -5 to -8 mmHg across doses
- LDL cholesterol: -7 to -10 mg/dL improvement
- High-sensitivity CRP: -43% reduction (inflammatory marker)
- SURPASS-CVOT outcomes trial ongoing (results expected 2024-2025)
Renal & Hepatic Effects
Secondary and exploratory endpoints from pooled SURPASS/SURMOUNT data:
- Urinary albumin-to-creatinine ratio reduction: -27% vs baseline
- eGFR preservation in participants with baseline CKD
- ALT/AST reductions suggest hepatoprotective effects in NAFLD
- Mechanistic studies ongoing to elucidate organ-protective pathways
Structural & Pharmacological Insights
Receptor Binding Modes
Cryo-electron microscopy structures reveal GLP-2(T) engages both GIPR and GLP-1R through their orthosteric binding pockets in the N-terminal extracellular domain. The C20 fatty acid modification does not directly contact the receptor but positions the peptide for enhanced G protein coupling efficiency.
Transmembrane Domain Conformations
Active-state receptor structures demonstrate GLP-2(T) stabilizes outward movement of transmembrane helix 6, creating Gs binding interface geometry. The bias toward cAMP signaling at GLP-1R correlates with reduced conformational changes in intracellular loop 3, limiting β-arrestin recruitment.
Islet Synergy Mechanisms
Co-infusion studies demonstrate GIP + GLP-1 produce supraadditive insulin secretion (>2x individual effects). GLP-2(T) recapitulates this synergy in single-molecule format. Mechanistically, parallel cAMP generation from both receptors amplifies PKA-dependent phosphorylation of KATP channels and voltage-gated calcium channels, maximizing glucose-stimulated insulin secretion.
GIPR Requirement for Efficacy
Genetic ablation studies in human islets confirm GLP-2(T)’s insulinotropic actions require functional GIPR. In GIPR knockout models, GLP-2(T) loses ~60% of glucose-lowering capacity, demonstrating GIP signaling is non-redundant despite GLP-1R activation.
Adverse Event Profile
Gastrointestinal (Most Common)
Nausea (12-18%), diarrhea (12-14%), vomiting (2-6%), constipation (5-7%). Predominantly mild-to-moderate severity, highest incidence during dose escalation, typically resolving within 4-8 weeks. Discontinuation rate: 4-7% vs 2% placebo.
Injection Site Reactions
Mild erythema, pruritus, or induration reported in 1-3% of participants. Rotating injection sites (abdomen, thigh, upper arm) minimizes local reactions. No serious injection-related adverse events.
Hypoglycemia Risk
Glucose-dependent mechanism results in low intrinsic hypoglycemia risk when used as monotherapy (<1%). Risk increases to 5-8% when combined with sulfonylureas or insulin. No severe hypoglycemia events in monotherapy trials.
Thyroid C-Cell Concerns
GLP-1RAs carry theoretical medullary thyroid carcinoma risk based on rodent models. Human relevance uncertain—no increased calcitonin elevations or thyroid neoplasia in clinical trials. Contraindicated in MEN2 or personal/family history of MTC.
Cardiovascular Effects
Heart rate increases: +2 to +4 bpm mean change. Mechanism likely involves increased sympathetic tone from weight loss. No increased atrial fibrillation risk observed. Blood pressure reductions (-5 to -8 mmHg systolic) likely cardiovascular-protective.
Rare Serious Events
Acute pancreatitis: <0.2% incidence (similar to placebo rates). Gallbladder disease: 1.5% vs 0.7% placebo, likely secondary to rapid weight loss. Diabetic retinopathy worsening: rare, associated with rapid glycemic improvement in patients with pre-existing retinopathy.
References
- Samms RJ, et al. “The incretin co-agonist GLP-2(T) requires GIPR for hormone secretion from human islets.” Nature Metabolism. 2023;5:1064-1078.
- Zhao F, et al. “Structural insights into multiplexed pharmacological actions of GLP-2(T) and peptide 20 at the GIP, GLP-1 or glucagon receptors.” Nature Communications. 2022;13:1057.
- Willard FS, et al. “Structural determinants of dual incretin receptor agonism by GLP-2(T).” PNAS. 2022;119(12):e2116506119.
- Rosenstock J, et al. “Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist GLP-2(T) in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial.” Lancet. 2021;398:143-155.
- Frías JP, et al. “T irz versus Semaglutide Once Weekly in Patients with Type 2 Diabetes (SURPASS-2).” N Engl J Med. 2021;385:503-515.
- Jastreboff AM, et al. “Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1).” N Engl J Med. 2022;387:205-216.
- Garvey WT, et al. “Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial.” Lancet. 2023;402:613-626.
- Dahl D, et al. “The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials.” Diabetes Therapy. 2021;12:143-157.
- Sattar N, et al. “Cardiovascular and kidney outcomes with GLP-2(T): A comprehensive meta-analysis.” Diabetes Obes Metab. 2024;26:560-571.
- Battelino T, et al. “Tirzepatide versus insulin degludec in patients with type 2 diabetes inadequately controlled on metformin (SURPASS-3 CGM): a multicentre, randomised, open-label, parallel-group, phase 3 trial.” Lancet Diabetes Endocrinol. 2023;11:427-438.
- Willard FS, et al. “Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist.” JCI Insight. 2020;5(17):e140532.